|
we assume that some linear combinations of atomic orbitals are stationary states). We can treat a molecular orbital as approximately a linear combination of two atomic orbitals, and solve the time-independent Schrodinger equation (i.e. The closest thing to a correct and intuitive explanation is the following: Is there a better explanation for why the interaction is the strongest between orbitals of similar energy? 
Wouldn't this make it, on average, non-bonding? However, if this were the case, then every molecular orbital made out of AOs of different energy would be bonding exactly half of the time, while being anti-bonding the other half. They can only be in phase all of the time if they have the same frequency, which is only possible if they have the same energy." So even if two orbital with identical shape and initial phase but different energy could exist, their overlap would only be constructive some of the time because they would not stay in phase. The frequency of this change is proportional to the energy of the orbital. "It's fairly common to think of orbitals as static, but their phase changes in time. 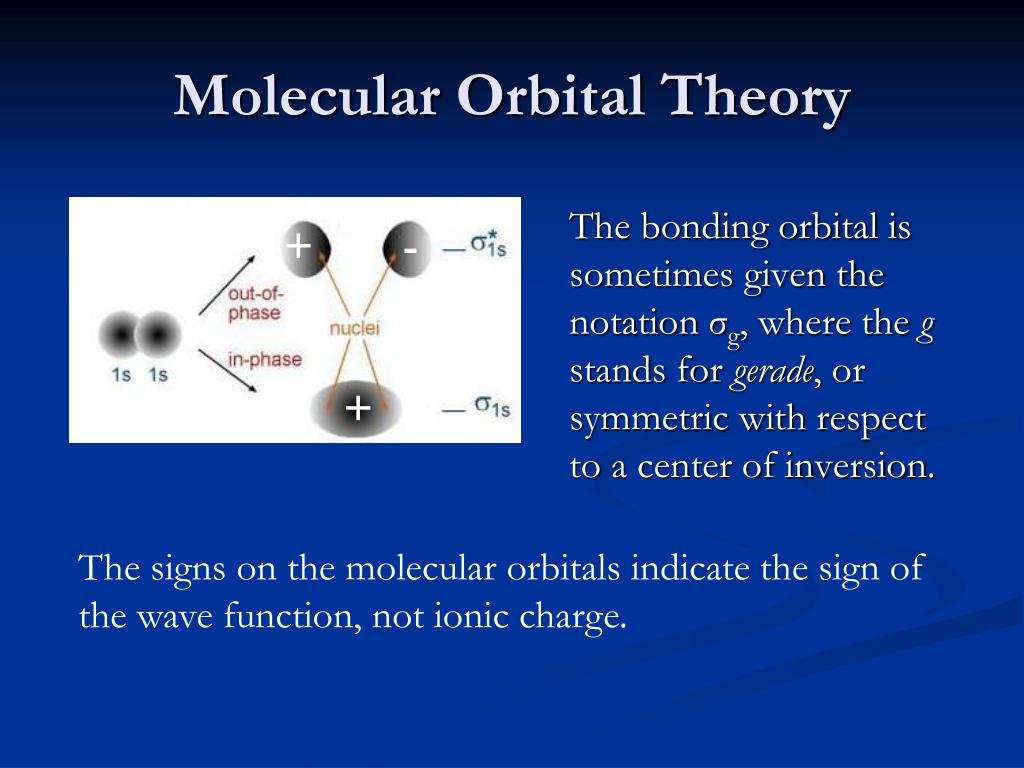
I understand the necessity for the same symmetry, but the only explanation for b) I've been able to find is this one: In molecular orbital theory, it is often said that only orbitals with a) the same symmetry and b) similar energy can interact to a significant degree.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
